1 / 5











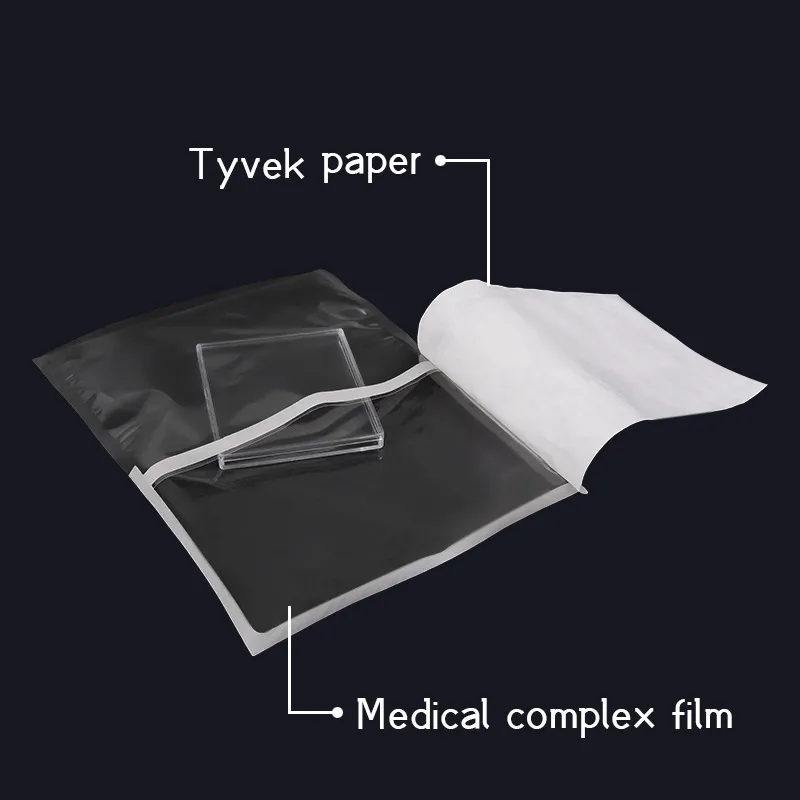
Medical Use Disposable Eo Sterilization Tyvek Header Pouch
| PROPERTY | SPECIFICATIONS |
|---|---|
| Product Name | Medical Use Disposable Eo Sterilization Tyvek Header Pouch |
| Material | Tyvek + Plastic |
| Usage | Medical Consumables Sterilization |
| MOQ | 10000pcs |
| Size Options | 45*58+15cm, 35*44+10cm, 23*36+8cm (Customizable) |
| Sterile Type | Sterile by Steam, EO, Gamma, Plasma, Electron Beam |
| Standards | ISO11607, EN868, YY/T 0698-9 |