1 / 5










| Model NO. | Marya AF | Machine Power | 1kw |
| Material | Stainless Steel | Control System | Automated Control |
| Sterilization | Vhp Generator | Compliance | GMP |
| Manufacturing Materials | 316L or PTFE | Origin | China |
| Production Capacity | 5000pieces/Year | Package Size | 340.00cm * 150.00cm * 200.00cm |
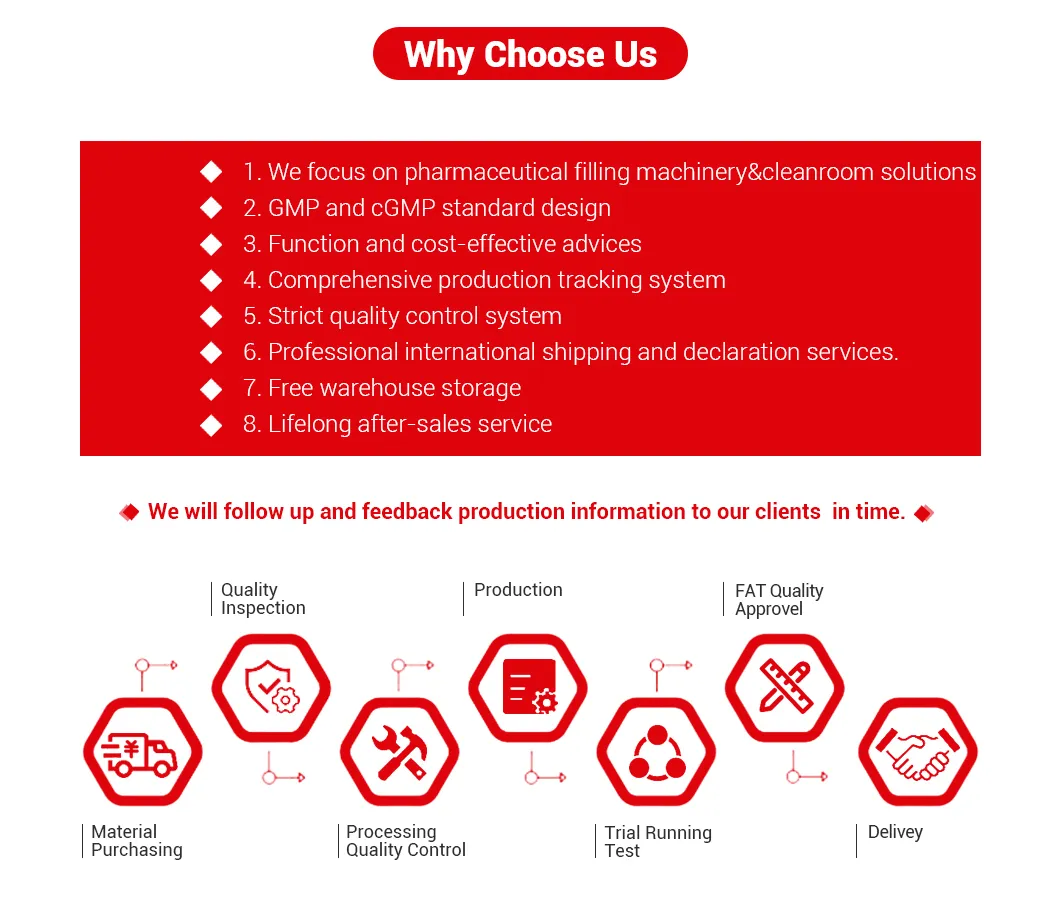
The Marya Advanced Flexible Isolation System delivers superior containment for critical aseptic pharmaceutical processes. This innovative system features modular, adaptable designs to meet diverse production needs while maintaining ISO 5/Class A environmental conditions.
Engineered with HEPA-filtered laminar airflow and ergonomic glove ports, it ensures optimal operator protection and product sterility. The system supports manual and automated operations, integrating seamlessly with filling lines for vaccines, biologics, and sterile injectables.


| Parameter Item | Specifications |
|---|---|
| Cleanliness | ISO 4.8 |
| Temperature and Humidity | Conventional 18-26ºC / 45%-65% (Customizable) |
| Differential Pressure | Regular 20-50pa |
| Air Flow Rate | 0.36m/s - 0.54m/s |
| Air Flow Type | Vertical one-way flow |
| Sterilization Effect | log6 and above |
| Leakage Rate | 0.5% |