Reduction in cross-contamination risks when switching from traditional stainless steel to single-use bio-disposable manifolds.
Elimination of CIP (Cleaning-in-Place) and SIP (Sterilization-in-Place) requirements, drastically cutting utility costs.
Faster speed-to-market for biopharmaceutical products using modular, pre-sterilized manifold assemblies.
In the contemporary biopharmaceutical landscape, Bio-disposable Manifolds have transitioned from a niche innovation to an industry standard. As the global demand for personalized medicine, vaccines, and monoclonal antibodies (mAbs) surges, manufacturers are facing unprecedented pressure to enhance flexibility while maintaining stringent sterility. Traditional fixed-piping systems are increasingly being replaced by custom-engineered single-use assemblies. These manifolds, typically comprised of medical-grade silicone or TPE tubing, connectors, and filters, provide a sterile, closed-loop environment that is critical for high-stakes bioprocessing.
The industrial shift is driven by the need for agility. Modern facilities must be capable of multi-product manufacturing, where rapid changeover is the key to profitability. Bio-disposable manifolds facilitate this by being "ready-to-use," eliminating the need for validation of cleaning cycles and reducing the footprint of massive sterilization infrastructure.
Post-pandemic, global pharmaceutical giants are re-evaluating their supply chains. There is a marked trend toward decentralized manufacturing, where localized production hubs require reliable, high-volume suppliers of disposable components. Procurement departments in the US, Europe, and SE Asia are looking for partners who can offer not just products, but validated technical expertise and supply chain resilience.
The industry is moving toward more robust materials. Modern bio-disposable manifolds must withstand extreme temperatures and maintain integrity under high-pressure conditions. We are seeing a shift toward gamma-stable materials that ensure no leachables or extractables contaminate the precious biological cargo. This level of material science is where top-tier Chinese manufacturers are now leading the charge.
Advanced manifolds are now integrated with RFID tags and sensors to track usage, sterilization dates, and flow data. This "Smart SUS" (Single-Use System) approach ensures that every component in the bioprocessing line is fully documented for regulatory compliance with agencies like the FDA and EMA.
As a premier China Bio-disposable Manifolds Manufacturer, Changzhou U-Care Medical Co., Ltd. bridges the gap between cost-efficiency and uncompromising quality. Located in the Yangtze River Delta, we leverage China's most advanced logistics and manufacturing ecosystem to serve clients worldwide.
Our modern production lines operate under strict ISO standards, utilizing Class 100,000 cleanrooms to ensure every manifold assembly meets the highest purity requirements of the medical sector.
We don't just sell parts; we provide solutions. Our R&D team works with your engineers to design custom manifolds with specific Y-connectors, T-pieces, and needle-free ports tailored to your process.
From raw material selection (USP Class VI) to final ETO or Gamma sterilization, every step is documented. Our "Quality First" ethos is backed by comprehensive testing protocols.
Changzhou U-Care Medical Co., Ltd. was established in 2014 and is located in Changzhou, Jiangsu Province, within the core region of the Yangtze River Delta—one of China’s most dynamic and economically advanced areas. Benefiting from a strategic location and convenient transportation network, the company enjoys strong access to both domestic and international markets.
U-Care Medical has accumulated extensive experience in the manufacturing of medical and pharmaceutical packaging products. The company is dedicated to the research, development, and production of high-quality disposable medical and pharmaceutical packaging materials. With a focus on innovation and reliability, U-Care strives to provide safe, efficient, and customized solutions to meet the evolving needs of healthcare and pharmaceutical industries worldwide.

We always place product quality at the highest priority. Customer satisfaction is the driving force behind our continuous improvement and development.
We are committed to building long-term partnerships with our clients by adhering to honest and transparent business practices, never compromising on ethics.
Through continuous innovation, we strive to develop more practical, reliable, and high-performance packaging solutions.
Our Vision: To become a trusted global provider of medical and pharmaceutical packaging solutions by consistently delivering superior quality, innovation, and customer-focused services.
Our Mission: To support the healthcare industry with safe, reliable, and efficient packaging products, contributing to better protection and delivery of medical products worldwide.

Our bio-disposable manifolds are utilized in diverse localized settings, adapting to specific regional regulatory and industrial requirements:
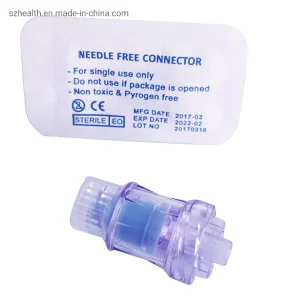
Infusion Needle Free Connection Valves Y-Port Valves Product
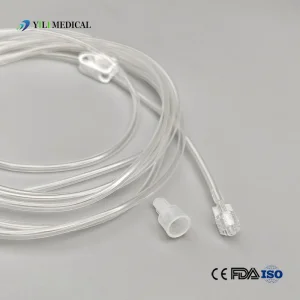
Customizable Silicone Tubing for OEM Peristaltic Pump Applications

High Barrier 11 Layer PA/EVOH/PE Multi-Layer Co-Extruded Film



Findou Oeb5 Isolator System for Versatile Pharmaceutical Solutions in China


Transfer Cleaning Assembly for Canon Imagerunner Advance C7055 C7065 C9065 PRO C9075 PRO