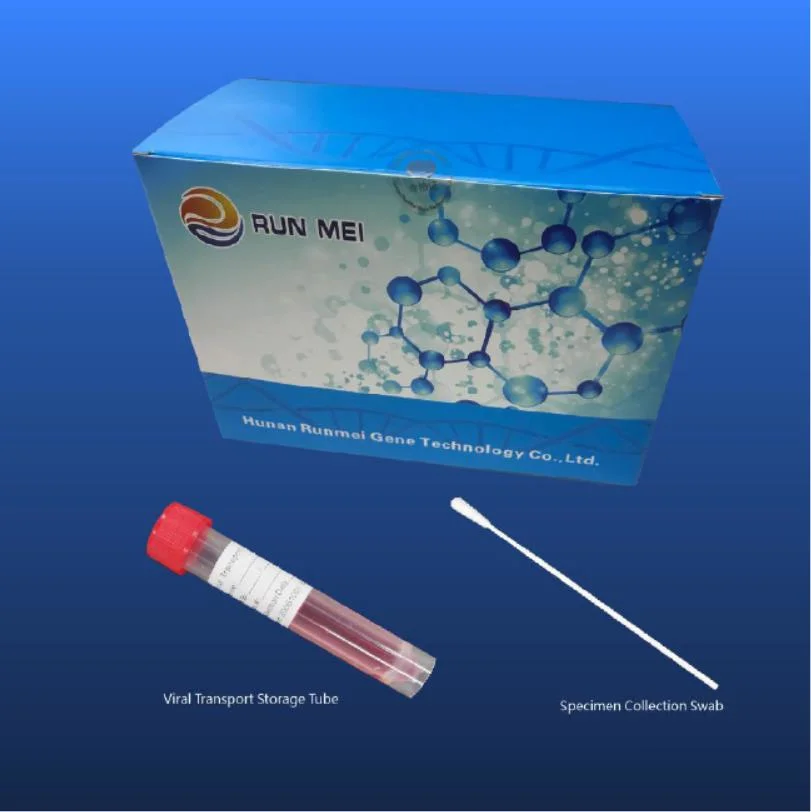
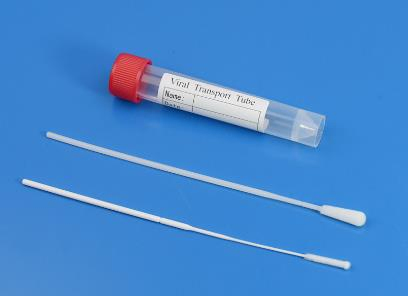
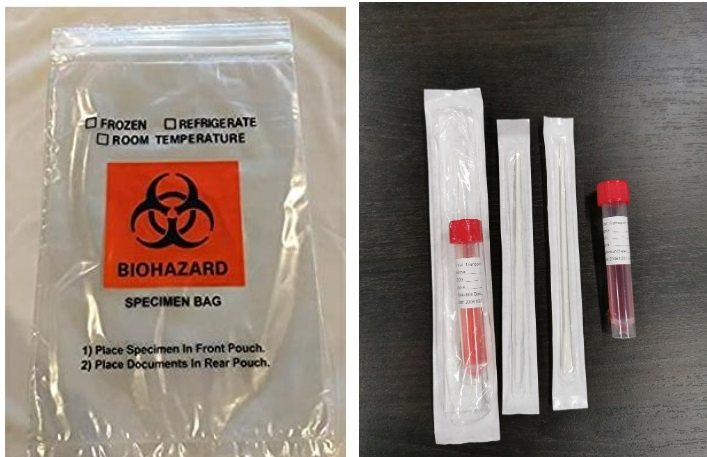












Sterile flocked throat forensic swab kit, medical disposable virus sampling with nasal swab kit, nylon nasopharyngeal swab FDA sampling tube with throat swab
Used for the collection of throat swab specimen and the next virus nucleic acid detection.
Suitable for any scene, no professional needed, no tedious operation.
Using multiple fluorescent PCR rapid detection technology, can detect three or more viruses at the same time, reducing detection costs and improving efficiency.
Using classic and accurate ELISA technology, on-site and portable monitoring of the immune effect.
As long as the reagents are purchased, the equipment can be supported with life-long after-sales service.
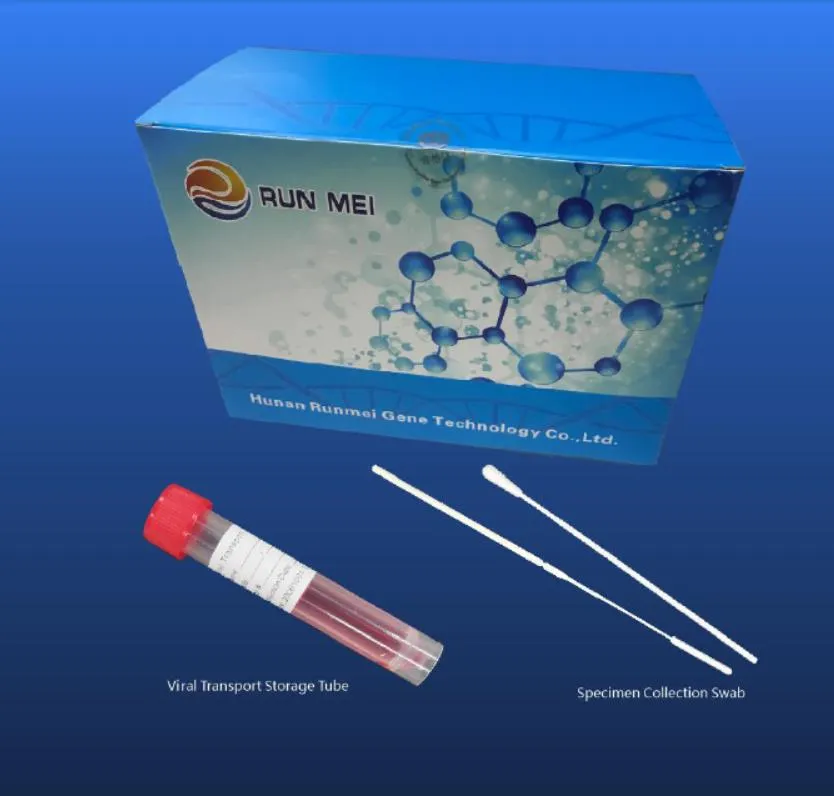
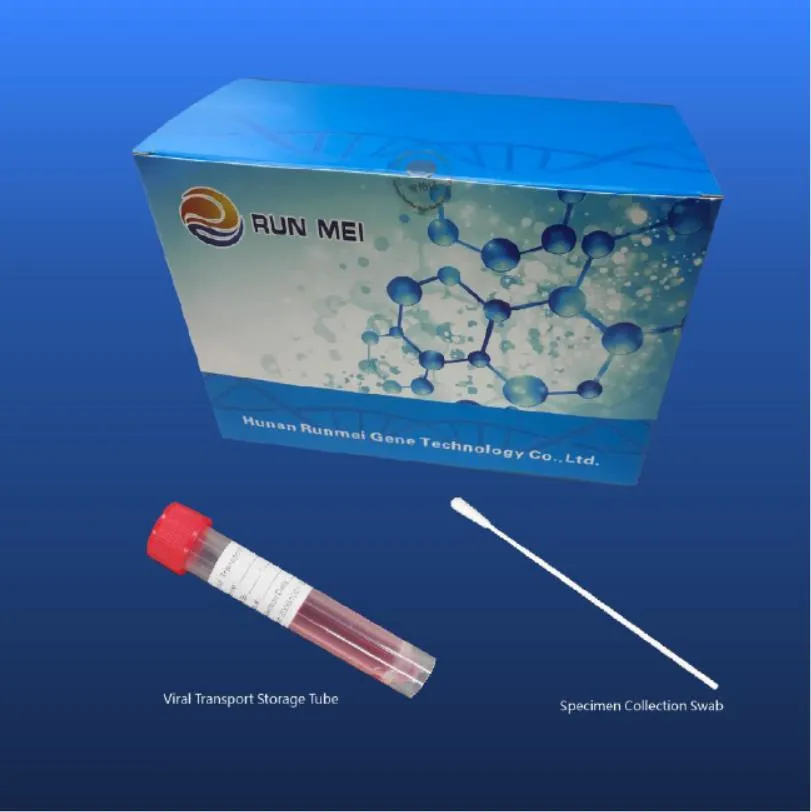
| No. | Swab Type | Tube | Viral Transport Medium | Intended Usage | Packing Specifications |
|---|---|---|---|---|---|
| 1 | One throat swab | One 10 ml tube | 3ml inactivated medium | Throat swab collection for nucleic acid detection. | 50 pcs/box; 20 box/carton; 13.6KG |
| 2 | One nasal swab | One 10 ml tube | 3ml inactivated medium | Nasal swab collection for nucleic acid detection. | 50 pcs/box; 20 box/carton; 13.6KG |
| 3 | One throat + one nasal | One 10 ml tube | 3ml inactivated medium | Combined collection for nucleic acid detection. | 50 pcs/box; 20 box/carton; 14.7KG |
| 4 | One throat + one nasal | Two 10 ml tubes | 3ml inactivated medium | Separate collection for nucleic acid detection. | 50 pcs/box; 20 box/carton; 24.2KG |
| 5 | One throat swab | One 10 ml tube | 3ml activated medium | Throat swab for virus isolation and culture. | 50 pcs/box; 20 box/carton; 13.6KG |
As a high-tech enterprise dedicated to the development of gene detection products and the construction of big data service platforms, we are led by a team of doctors. Our strategic goal is to solve the pain points and difficulties of the industry and create value for human beings. At present, the construction of product systems for pathogen biology fluorescence quantitative PCR detection kits, ELISA detection kits, and immune colloidal gold detection kits has been completed.






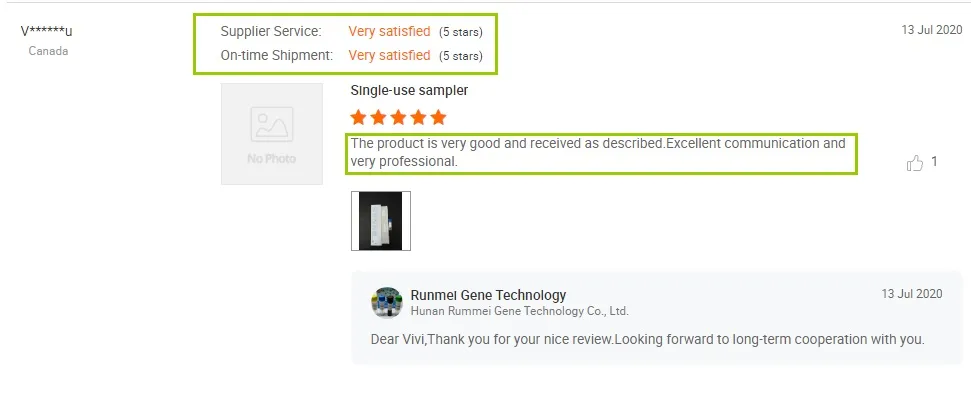
Customer Reviews: