Product Description

1Product Introduction
Our liquid preparation system normally includes various specialized configurations to meet pharmaceutical standards:
- Oral liquid preparation system: Includes sugar melting, initial preparation, cold storage, filtration, fine preparation, and CIP/SIP control.
- Sterile preparation system: Designed for large/small volume injections, eye drops, and water injections with aseptic storage and filtration.
- Extraction preparation system: Features enrichment, centrifugation, and thermal management for extraction processes.
- Cream and ointment system: Specialized in homogenization, emulsification, and aqueous/oil phase management.
Sterility & Quality Control
The system adopts closed tanks, aseptic connectors, and online Sterilization (SIP) functions. High-precision weighing modules and mass flowmeters ensure the accuracy of material feeding, while online monitoring of pH, conductivity, and temperature reduces manual errors.
Automation & Compliance
Complies with FDA 21 CFR Part 11 and ALCOA+ principles. Features PLC/SCADA control for automated execution of formula steps and remote monitoring for predictive maintenance.
Company Profile & Advantages

We are a leading pharmaceutical equipment manufacturer specializing in energy-efficient liquid preparation systems. Our solutions comply with FDA, cGMP, and QS standards, serving industries such as biopharmaceuticals, medical aesthetics, and food production.

Key Advantages
- Certifications: ISO9001, CE, and GC2 pressure vessel qualifications.
- Technological Edge: Modular design using 316L stainless steel for high corrosion resistance.
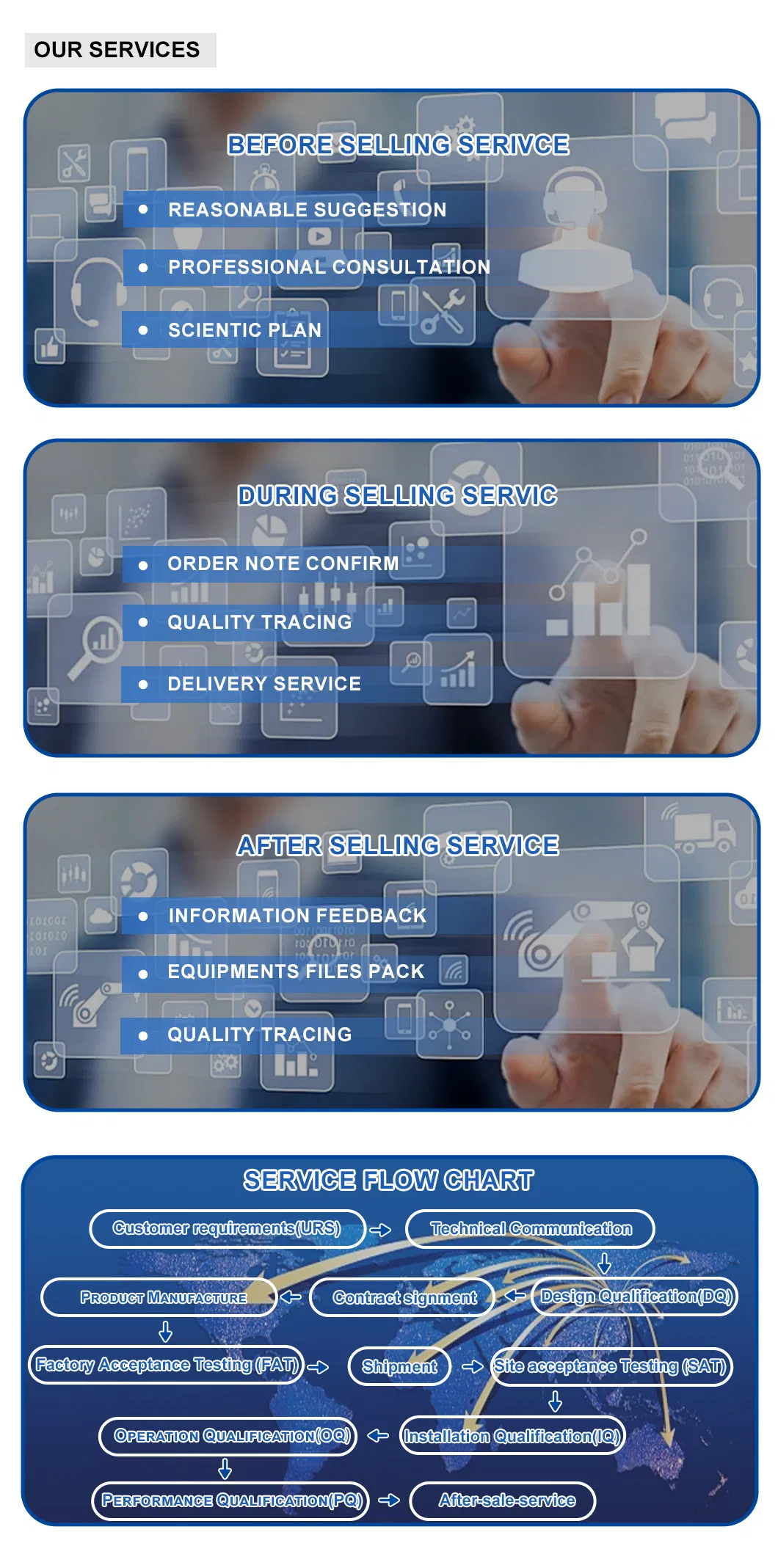
- Full Validation: Provision of complete validation documents (URS, FAT, SAT, DQ, IQ, OQ, PQ).
- Expert Support: 24-hour rapid response and technical training services.
Frequently Asked Questions
Q1: Does your equipment comply with international standards?
Yes, all equipment is designed and manufactured according to GMP, FDA, and WHO requirements to ensure global compliance.
Q2: What materials are used in the preparation systems?
We primarily use 304 and 316L stainless steel for all contact parts, ensuring high corrosion resistance and hygiene.
Q3: What is the typical lead time for an order?
Standard systems usually take 4-6 weeks, while fully customized liquid preparation lines may take 6-12 weeks depending on complexity.
Q4: What kind of after-sales technical support do you provide?
We offer 24/7 technical support via remote troubleshooting, video calls, and if necessary, on-site engineer visits for maintenance.
Q5: How are the systems packaged for international shipping?
We use heavy-duty reinforced wooden packages to ensure the safety of the equipment during sea or air freight.
Q6: Do you provide validation documentation?
Yes, we provide a full set of validation documents including URS, FAT, SAT, DQ, IQ, OQ, and PQ to assist with your certification process.