




1 / 5










1. Description of the preparation system:
The preparation system is a type of pharmaceutical equipment used to mix and dispense liquids by adding an API to a solvent such as water for injection and then formulating it into a pharmaceutical agent that meets the desired concentration after stirring, heating, sterilization, and filtration.
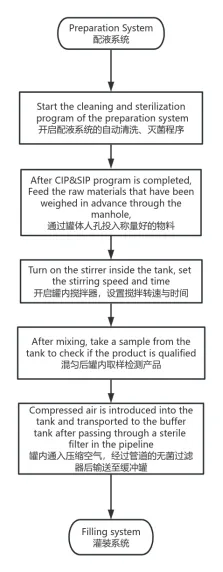
2. Classification of the preparation system:

General preparation system: Used for general small molecule chemical preparation, such as aqueous injection and lyophilized powder injection.

Complex preparation system: Used for suspensions, emulsions, liposomes, and microspheres.

Biological preparation system: Ideal for antibodies, vaccines, blood products, and recombinant proteins.

| Component Name | Technical Parameters |
|---|---|
| Working volume | 2 ~ 15000L |
| Stirring speed accuracy | 0 ~ 450rpm ±3.0% |
| Temperature control accuracy | -10 ~ 150ºC ±0.2ºC |
| Pressure control accuracy | -0.01Mpa ~ 0.06Mpa ±0.01MPa |
| Weighing control accuracy | 3‰ |
| Power supply | 380V 50Hz (Other voltages customizable) |
| Protection level | IP65 |












